Our Research
Inherited retinal degenerative diseases are a major cause of blindness worldwide. In these conditions, photoreceptors, the cells responsible for transforming light into a signal that can be transmitted to and interpreted by the brain, are dysfunctional and/or slowly destroyed.
Veterinary ophthalmologists and vision scientists from the Division of ExpeRTs are dedicated to identifying the genetic causes of inherited blindness, delineating the mechanistic basis of genetic visual defects, and developing innovative and effective treatments to preserve or restore sight to the visually impaired.
Through the use of canine models, the group has contributed to a better understanding of the molecular mechanisms leading to disease, and is contributing to the development of novel therapies for both dogs and people.

Discovery of Novel Genes Causing Retinal Degeneration in Dogs
Over a hundred different breeds of dogs are affected with inherited forms of retinal degeneration that lead to progressive vision loss.
Two general approaches are used to identify genes associated with inheritable visual defects:
- A “candidate gene approach” where genes are selected based on their expected contribution to given disease characteristics
- A “genome wide screening approach” utilizing informative pedigree resources to identify the disease-bearing chromosomal region, followed by cloning and identification of the causative mutation
There are currently close to 45 different genes known to carry mutations responsible for retinal degeneration in dogs. Genetic studies carried out by Division of ExpeRTs investigators have identified mutations in many of these genes, and developed genetic tests that are helping veterinarians and dog breeders establish a molecular diagnosis and prevent these blinding diseases.
Deciphering the Cellular and Molecular Mechanisms of Photoreceptor Disease
Vision scientists at the Division of ExpeRTs are actively investigating the cascade of cellular and molecular events that are triggered in the retina by genetic mutations and environmental factors, and that subsequently lead to disease and death of rods and cones, the two classes of photoreceptor cells that are critical for vision.
Analysis of specific pathways of cell death or cell survival enables identification of potential molecular targets for development of new therapeutic strategies. For example, identification of decreased levels of an anti-death gene called X-linked inhibitor of apoptosis (XIAP) in the degenerating retinas of dogs, has prompted our group to investigate whether XIAP supplementation via gene therapy can halt the death of photoreceptors.
Using state of the art transcriptomic technology, Division of ExpeRTs investigators have also discovered that numerous molecular signaling pathways of inflammation are activated in early and late stages of retinal disease in dogs, prompting to investigate the role of anti-inflammatory therapeutics in these neurodegenerative conditions.


Canine Models
While the value of rodent models in vision research is undisputed, the complementary use of canine models offers many unique advantages.
In particular, the anatomy of the canine and human eye is quite similar and the presence of a recently discovered cone-rich central retinal region (the canine “fovea-like area”), a well developed retinal vasculature, and comparable size advances the transfer of therapies developed in canine models to human patients.
Investigators from the Division of ExperRTs have studied more than 20 naturally-occurring canine diseases, most of which share striking similarities with their human equivalent, thus providing naturally-occurring models of Leber congenital amaurosis, early and late onset retinitis pigmentosa, achromatopsia (day blindness), cone rod dystrophy, and bestrophinopathy.
For further details please contact us.
Translational Retinal Therapies
Investigators at the Division of ExpeRTs have, in collaboration with other academic and industry scientists, pioneered the development of novel treatments for canine retinal diseases and their translation into several human clinical trials.
By using these canine models, they are able to evaluate in a human-sized eye the ability to successfully deliver drugs to the retina, demonstrate structural and functional rescue of retinal cells, preservation or restoration of visual cortical activity, and visually-guided behavior.
Following successful outcome with these proof of concept studies the Division of ExpeRTs is able to support non-clinical IND-enabling studies.
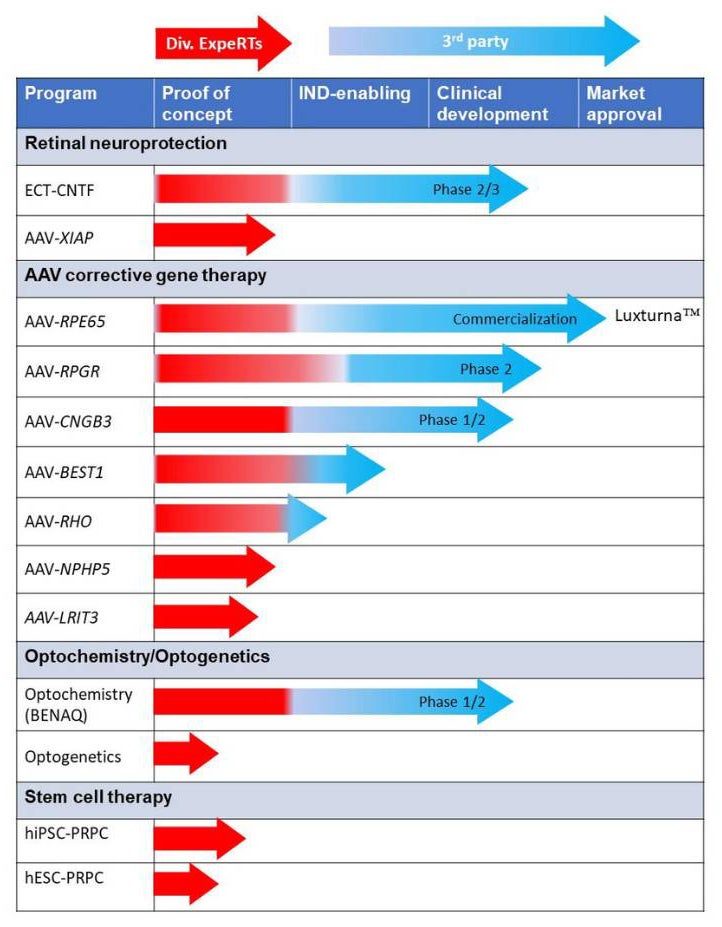
Investigators at the Division of ExpeRTs were the first to test in a canine model of retinal degeneration, an encapsulated cell therapy (ECT) device aimed at providing sustained intravitreal delivery of a prosurvival protein called ciliary neurotrophic factor (CNTF).
The study demonstrated that the ECT-CNTF device could slow disease progression and rescue photoreceptors, and the technology was subsequently moved to clinical trials for patients affected with retinitis pigmentosa (RP), geographic trophy, and macular telangiectasia.
More, recently, the Division of ExpeRTs have been investigating another neuroprotective strategy that consists in delivering via an adeno-associated virus (AAV) an anti-cell death gene (X-linked inhibitor of apotosis; XIAP) that has show to confer rescue in several rodent models of retinal degeneration.
Division of ExpeRTs investigators have a long-term expertise in AAV vector-mediated retinal gene therapy. In collaboration with other teams, they have to this date demonstrated positive outcomes in canine models of six human diseases, three of which were successfully translated to clinical trials, and the other three are in the IND-enabling phase.
Identification of a naturally-occurring mutation in the RPE65 gene in Briards, a breed of sheepdogs predisposed to a form of congenital blindness similar to human disease, provided a large animal model for testing gene therapy approaches to cure blindness. Gene therapy in the dogs was successful and Lancelot, a dog with restored vision, became a popular “spokesdog” for increasing public awareness of the potential of gene therapy. The group’s pioneering work on this canine model led to several clinical trials in patients with Leber congenital amaurosis (LCA), and culminated in 2018 with the commercialization in the US and Europe of the first retinal gene therapy for patients affected with the RPE65 form of LCA or RP.

AAV-RPE65 is only one of the many gene therapy programs that has been led by investigators at the Division of ExpeRTS and include:
- Gene therapy for a form of congenital stationary night blindness (CSNB) caused by mutations in the LRIT3 gene.
- Gene therapy for a form of day blindness (called achromatopsia) caused by mutations in the CNGB3 gene.
- Gene therapy for the most common and severe form of X-linked retinitis pigmentosa caused by mutations in the RPGR gene.
- Gene therapy for the most common form of autosomal dominant retinitis pigmentosa caused by mutations in the rhodopsin (RHO) gene.
- Gene therapy for an inherited form of macular degeneration caused by mutations in the bestrophin (BEST1) gene.
- Gene therapy for a form of LCA caused by mutations in the ciliopathy gene IQCB1/NPHP5.
In collaboration with other academic and industry partners, the Division of ExpeRTs is investigating the use of optogenetic tools and small photoswitch molecules to restore vision when retinas are treated at a stage when all photoreceptors (rods and cones) have already died. These strategies aim at rendering other neuronal cell populations (that survive even in advanced degeneration) sensitive to light stimuli, with the goal of restoring some visual perception.
As an other approach to restore vision in animals or people with end stage retinal degeneration the Division of ExpeRTs is evaluating the delivery of stem cell-derived (ESC and iPSC) photoreceptor-precursor cells as a way of replacing rods and cones.


