Penn Vet Researchers Uncover a Shared Cellular Stress Signal that May Drive Vision Loss in Retinitis Pigmentosa
Retinitis pigmentosa (RP) is a group of inherited eye diseases that cause progressive vision loss through the degeneration of photoreceptors, the retina’s light-sensing cells. Although mutations in more than 100 genes are linked to RP, the cellular stress pathways that ultimately cause photoreceptor death remain incompletely understood, limiting the development of broadly applicable therapies.
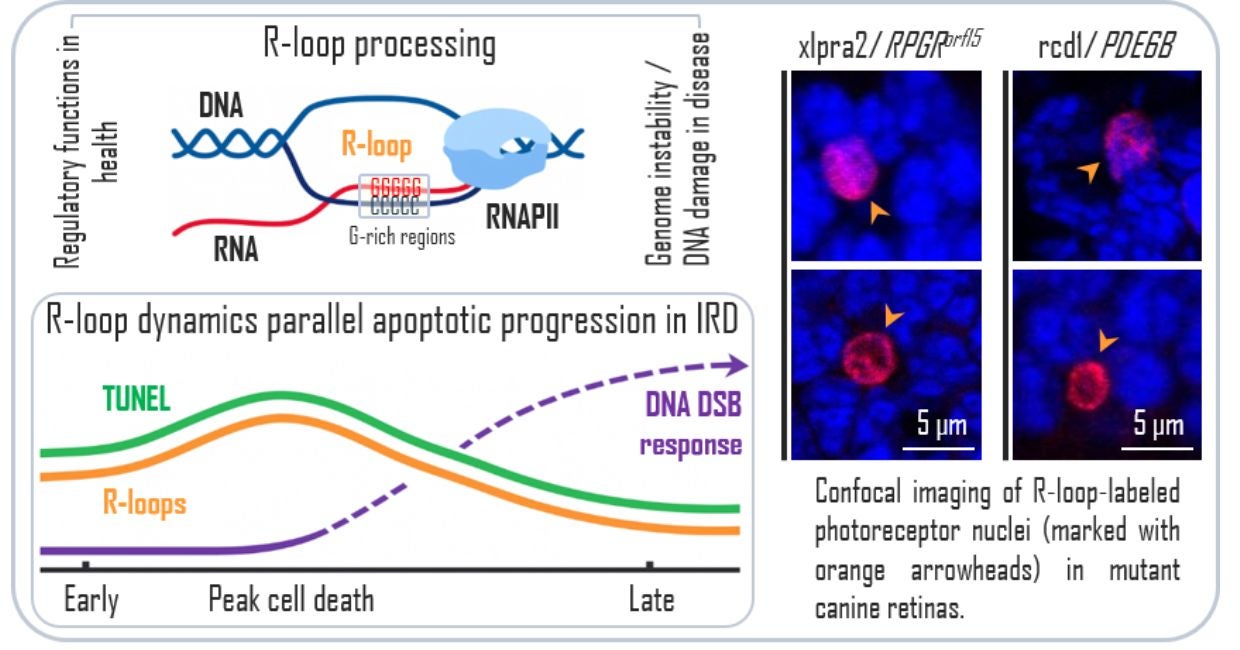
In a new study published in Investigative Ophthalmology & Visual Science, vision scientists from the Division of Experimental Retinal Therapies (ExpeRTs) at the University of Pennsylvania’s School of Veterinary Medicine report abnormal accumulation of R-loops in photoreceptor nuclei in two naturally occurring canine models that closely mirror human RP.
R-loops are short-lived RNA-DNA three-stranded structures that form when newly synthesized RNA binds to its DNA template, leaving the opposite DNA strand temporarily unpaired. This is like when a zipper gets caught with fabric. Under healthy conditions, R-loops are carefully regulated by the cell and help genes function properly, including making and processing RNA and maintaining DNA stability. When this control breaks down, too many R-loops can form, damaging DNA and making the genome less stable.
This study is the first to examine whether the harmful buildup of R-loops is linked to inherited retinal diseases. Using two dog models of early-onset retinitis pigmentosa, the researchers studied vision loss caused by different gene mutations. Even though the genetic causes differed, both models showed similar molecular changes as the disease worsened.
“We found that most of the photoreceptors that were dying also showed unusually high levels of R-loops, along with changes to RNA that are known to affect how cells manage stress and control cell death,” says Tatyana Appelbaum, PhD, the study’s lead author and a senior research investigator. “Although this does not prove cause and effect, seeing the same pattern in two different disease models suggests there may be common stress responses that make these cells more vulnerable.
The team also observed that R-loop levels rose and declined in parallel with the progression of cell death in both models. Near the peak of photoreceptor degeneration, the researchers detected altered expression of genes involved in R-loop regulation, along with signs of abnormal RNA processing, the system cells use to correctly read and manage genetic instructions. At later disease stages, R-loop levels declined while markers of DNA damage increased, suggesting a shift in cellular stress responses.
“These findings suggest that abnormal R-loop accumulation, likely arising from impaired RNA processing, represents a shared stress response in photoreceptor degeneration, even when the underlying genetic mutations are very different,” says William A. Beltran, DVM, PhD, DECVO, the Corinne R. and Henry Bower Professor of Ophthalmology, Director of the Division of ExpeRTs, and senior author of the study. “Identifying a common molecular mechanism of stress or death across a genetically diverse group of inherited blinding diseases may pave the way to finding a treatment that could be administered irrespective of the type of genetic mutation that triggers the condition. This would have a huge impact in the field as there are more than 100 genes that cause a form of retinitis pigmentosa.”
Looking ahead, the team plans to investigate how R-loop dysregulation emerges across different forms of retinal degeneration and whether stabilizing these downstream stress responses, despite distinct genetic causes, could help protect vulnerable photoreceptors.
“Targeting R-loop homeostasis represents a potential therapeutic direction,” Appelbaum says. “Our study provides a foundation for exploring whether stabilizing these pathways could slow disease progression in inherited retinal diseases.”
This work was supported by the NIH/National Eye Institute and the Foundation Fighting Blindness.
Related News

New Stamps Scholars Program at Penn Vet to Support Exceptional VMD and VMD-PhD Students
The University of Pennsylvania School of Veterinary Medicine (Penn Vet) has partnered with the Stamps Scholars Program to establish the Stamps VMD/VMD-PhD Fellows, a prestigious scholarship initiative recognizing exceptional veterinary…

Anthony Fauci Delivers Robert R. Marshak Keynote at Penn Vet’s 2026 Institute for Infectious and Zoonotic Diseases Symposium
The University of Pennsylvania School of Veterinary Medicine’s (Penn Vet) Institute for Infectious and Zoonotic Diseases (IIZD) convened leading experts in infectious disease research for its 2026 annual symposium, held…

A Magical Discovery Reaches Across the Waves, the Years, and the Miles
Greg Lewbart, V’88, and his wife Diane Deresienski, V’89, were leading a research expedition in the Galapagos Islands when a member of their party, Andrea Loyola, head veterinarian for the…
About Penn Vet
Ranked among the top ten veterinary schools worldwide, the University of Pennsylvania School of Veterinary Medicine (Penn Vet) is a global leader in veterinary education, research, and clinical care. Founded in 1884, Penn Vet is the first veterinary school developed in association with a medical school. The school is a proud member of the One Health initiative, linking human, animal, and environmental health.
Penn Vet serves a diverse population of animals at its two campuses, which include extensive diagnostic and research laboratories. Ryan Hospital in Philadelphia provides care for dogs, cats, and other domestic/companion animals, handling more than 30,000 patient visits a year. New Bolton Center, Penn Vet’s large-animal hospital on nearly 700 acres in rural Kennett Square, PA, cares for horses and livestock/farm animals. The hospital handles more than 6,300 patient visits a year, while our Field Services have gone out on more than 5,500 farm service calls, treating some 22,400 patients at local farms. In addition, New Bolton Center’s campus includes a swine center, working dairy, and poultry unit that provide valuable research for the agriculture industry.

