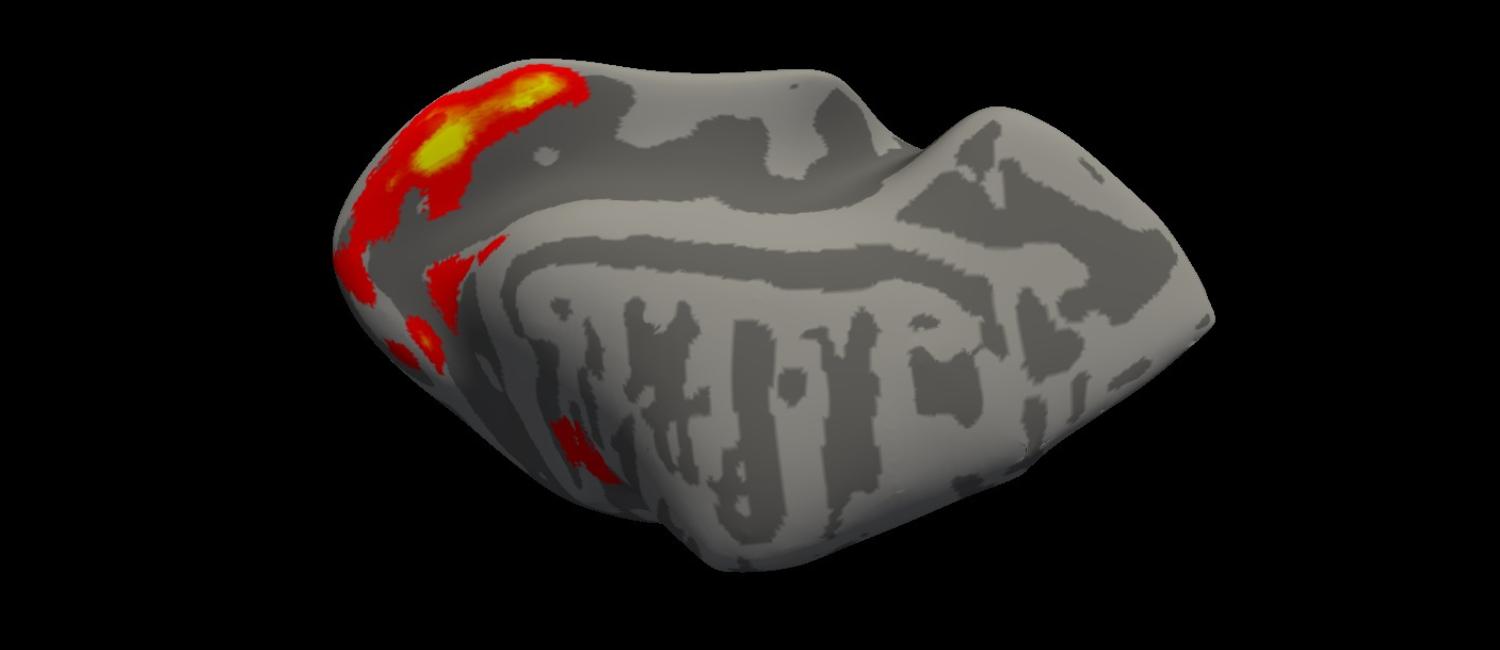
 Illustration of fMRI activity in the visual area of a dog brain, inflated to show the sulci and gyri of the brain. (Image: Courtesy of Huseyin O. Taskin)
Illustration of fMRI activity in the visual area of a dog brain, inflated to show the sulci and gyri of the brain. (Image: Courtesy of Huseyin O. Taskin)
In the retinas of human eyes, the cones are photoreceptor cells responsible for color vision, daylight vision, and the perception of small details. As vision scientists from the Division of Experimental Retinal Therapies at the University of Pennsylvania School of Veterinary Medicine, Gustavo D. Aguirre and William A. Beltran have been working for decades to identify the basis of inherited retinal diseases. They previously showed they could recover missing cone function by reintroducing a copy of the normal gene in photoreceptor cells.
Both humans and dogs are affected by retinal disease, and a new study of daylight vision using a canine model offers a critical insight for evaluating “whether these cell replacements—where we are introducing cones into the retinas of these dogs—is a successful approach for restoring cone vision,” says Beltran, the Corinne R. and Henry Bower Endowed Professor of Ophthalmology.
He and Gustavo Aguirre teamed with researchers including cognitive neuroscientist Geoffrey K. Aguirre, a professor of neurology at the Perelman School of Medicine, bringing together knowledge on the retinal system and brain measurements. In dogs with three different kinds of naturally occurring retinal disease and in dogs with normal vision, the scientists used functional magnetic resonance imaging (fMRI) to assess brain responses to lights that stimulate only the cone cells.
The researchers found that fMRI can detect brain responses to daylight vision for black and white information as well as color information, and it can identify the area of the visual cortex that responds to stimulation of a region in the dog retina that is rich in cones and similar to the human fovea. They also found they can use fMRI to measure the relative degree of loss of daylight vision. Using this technique in animals with a retinal disease caused by a mutation in a gene called NPHP5, they demonstrated that gene augmentation therapy restored the response in the cortex to black and white stimulation. That makes this disease a promising one in which to study photoreceptor cell replacement as a treatment in the future.
Their findings were published in Translational Vision Science & Technology. The other co-authors are Huseyin O. Taskin, a former research specialist at Penn in the GKAguirre Lab and current graduate student at the University of Toronto, and Jacqueline Wivel, a veterinary technician.
“Canine models are useful for studying retinal diseases because they have a variety of different naturally occurring genetic disorders. The ultimate goal is to first demonstrate that these disorders can be treated in canines before translating it to human patients,” says Taskin, the first author. Gustavo Aguirre says, “The hope is that successful therapeutic approaches in people will then become available to veterinarians so that they can benefit man’s four-legged friend.”
Geoffrey Aguirre says, “The purpose of the study was to see, in different versions of these retinal diseases, how much information about daylight vision makes it to the visual system in these dogs.” This knowledge is particularly useful, he says, because figuring out whether a treatment for retinal disease has been effective requires knowing how much vision function was present prior to treatment.
Beltran says this paper shows that gene therapy can recover cone function because it looks at an animal model with no cone function and shows an improvement. He explains that, in the disease caused by the NPHP5 mutation, cones are present but not functional. Animals with this disease are born day-blind but initially have some night vision, though rods—photoreceptors that allow night vision—die over a period of months, making dogs fully blind within a year.
Previous research has measured responses to stimuli through electroretinography and visual behavior tests, which Beltran says can require weeks if not months of dog training. Geoffrey Aguirre says the use of fMRI in this study is significant because it is faster and easier than measuring behavior, and it is not invasive. Taskin also notes that neither visual behavior tests nor electroretinography provide certainty as to what happens in the visual cortex.
A prior study showed that retinal gene therapy in a type of blinding disorder called Leber congential amaurosis is associated with restoration of fMRI responses from the canine visual cortex, but the nature of that study meant that both rod and cone responses could have contributed to cortical activity. The new study furthers knowledge of retinal disease by specifically stimulating the cones.
Huseyin O. Taskin is a former research specialist in the Department of Neurology at the Perelman School of Medicine and a current doctoral candidate in medical sciences at the University of Toronto.
Jacqueline Wivel is a certified veterinary technician in the University of Pennsylvania School of Veterinary Medicine.
Gustavo D. Aguirre is professor of medical genetics and ophthalmology in Penn Vet’s Department of Clinical Sciences & Advanced Medicine.
William A. Beltran is the Corinne R. and Henry Bower Endowed Professor of Ophthalmology and director of the Division of Experimental Retinal Therapies in Penn Vet’s Department of Clinical Sciences & Advanced Medicine.
Geoffrey K. Aguirre is professor of neurology, associate director of the Penn Center for Neuroscience and Society, and associate director of neurology residency in the Department of Neurology at Penn Medicine.
This study was supported by the National Eye Institute (grants R24-EY029890, RO1-EY006855, RO1-EY017549, and P30-EY001583), Fighting Blindness Canada Vision 20/20, The Foundation Fighting Blindness, and the Low Vision Research Award from the Research to Prevent Blindness / Lions Clubs International Foundation.